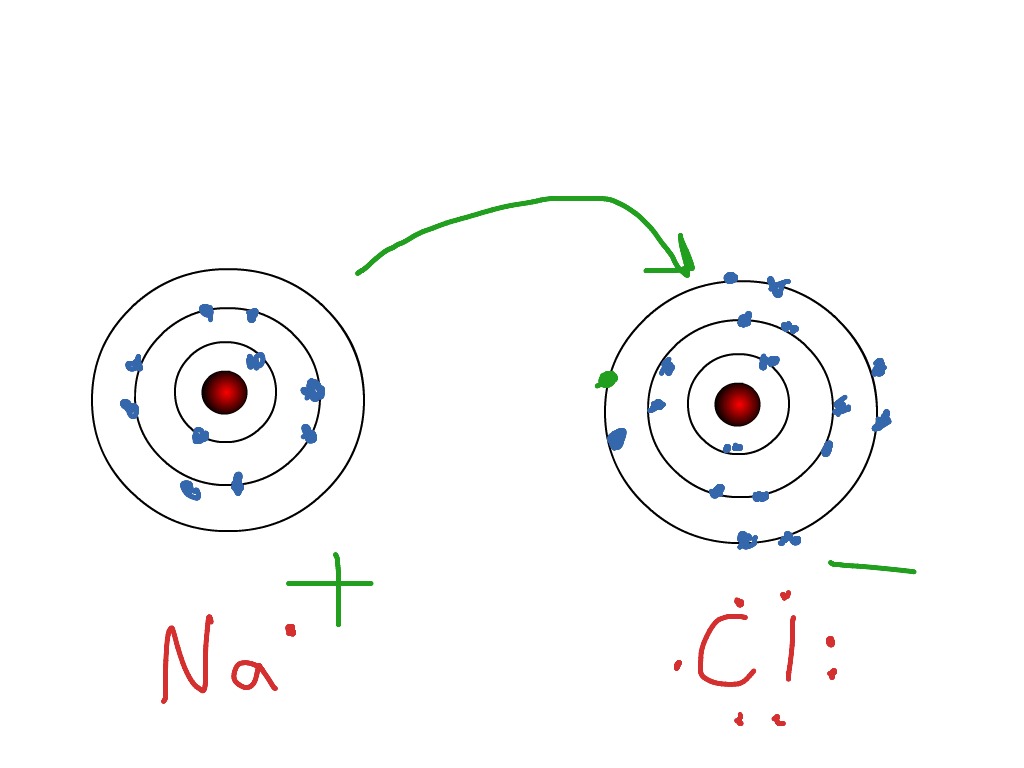
Electrovalence, as opposed to covalence, refers to the transfer of electrons.Ĭlean ionic bonding, in which one atom or molecule entirely transfers an electron to another, is impossible to achieve: all ionic compounds have some degree of covalent bonding or electron sharing. Ions are positively charged atoms that have lost their electrons (called cations). Negatively charged ions are formed when atoms gain electrons (called anions). Ions are electrostatically charged atoms (or groups of atoms). Along with covalent and metallic bonding, it is one of the most common types of bonding. Ionic bonding is the principal interaction happening in ionic compounds, and electrostatic attraction between different type of charged ions or between two atoms with significantly dopposite electronegativities is involved. A molecule is a chemical’s smallest unit.In a molten state, however, the electrostatic forces of attraction between the ions are overcome by the heat emitted, making them good conductors.Ionic compounds conduct zero electricity in the solid-state due to the impossibility of ion mobility, but they do so in the liquid state.Ionic compound solubility: Ionic compounds are normally soluble in polar solvents like water, but their solubility decreases in non-polar solvents like petrol and gasoline.As a result, they have high boiling and points.Ionic compound melting and boiling points: Breaking the ionic bonds between the atoms requires a lot of energy.As a result, they’re regarded as brittle. When pressure is applied, however, they disintegrate.Because of the strong attraction between negative and positive ions, ionic compounds are difficult to break.Ionic compounds have the following properties: As a result, ionic compounds have high boiling and melting points. Because the ions are securely bound in these massive complexes, breaking all of the connections takes a lot of energy. The ionic connections between the charged particles from a massive ion structure. The forces of attraction between the ions keep the ions in place. Because the quantity of sodium ions equals the number of chloride ions, the crystal is uncharged. As a result, a three-dimensional structure of Na+ and Cl– ions alternates. Sodium ions, for example, attract chloride ions, and chloride ions attract sodium ions. solid ions are held together by the electrostatic contact between positive and negative ions. Ionic compounds include oxides, salts, hydroxides, sulfides, and a vast variety of inorganic compounds. The structure of an ionic molecule is determined by the comparative sizes of the anions and cations. They are almost always electrically insulating as solids, but when melted or dissolved, the ions are mobilized, and they become extremely conductive.īecause oppositely charged ions are present, the electrostatic force of attraction holds ionic compounds together firmly. Ionic compounds are often rigid and brittle, with high melting and boiling temperatures. Evaporation of the solvent, precipitation, freezing, a solid-state reaction, or the electron transfer reaction of reactive metals with reactive non-metals, such as halogen gasses, can all be used to make ionic compounds from their constituent ions. Salts are ionic substances that lack these ions and can be produced by acid-base processes. When solid, ionic substances usually form crystalline forms.īases are ionic substances that include the basic ions hydroxide (OH) or oxide (O2). Individual ions in an ionic compound frequently have many nearest neighbors, hence they are not considered molecules but rather part of a three-dimensional network. 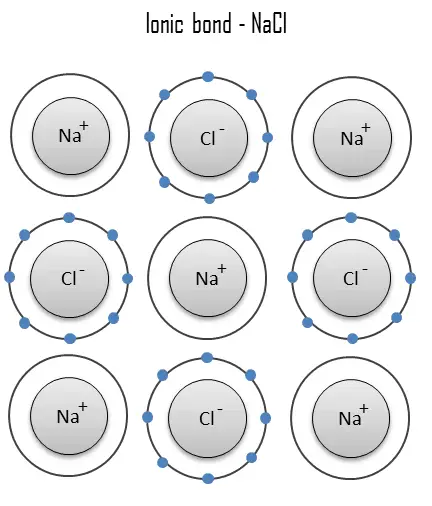
Simple ions like sodium (Na+) and chloride (Cl-) in sodium chloride, or polyatomic species like ammonium (NH+ 4) and carbonate (CO²-3) ions in ammonium carbonate, are examples. Overall, the chemical is neutral, although it contains both positively and negatively charged ions known as cations and anions. Ionic compounds:Īn ionic compound is a chemical complex made up of ions kept together by electrostatic forces known as ionic bonding in chemistry. 
A 2+ charged cation will form a stronger ionic connection than a 1+ charged cation.Īn ionic compound is a chemical complex made up of ions kept together by electrostatic forces known as ionic bonding in chemistry. 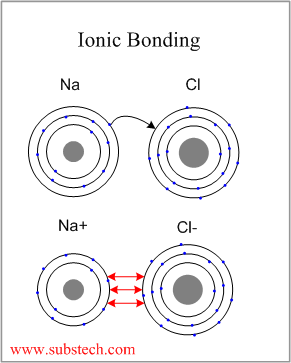
The ionic bond’s strength is proportional to the number of charges and inversely proportional to the distance between the charged particles. An ionic bond is a name for this type of relationship. The chemical bond is formed when electrons are transferred from one atom to another.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
